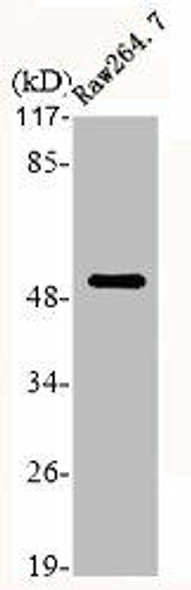
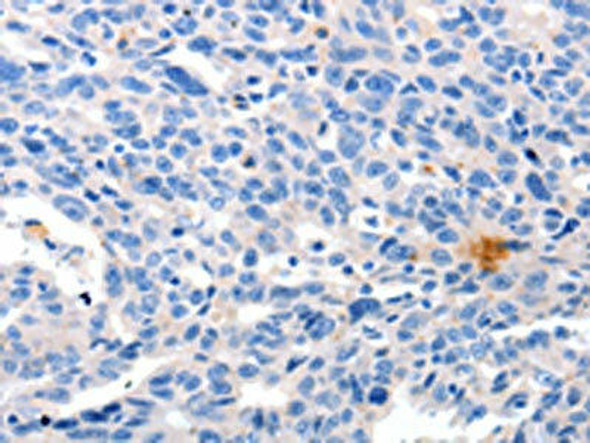
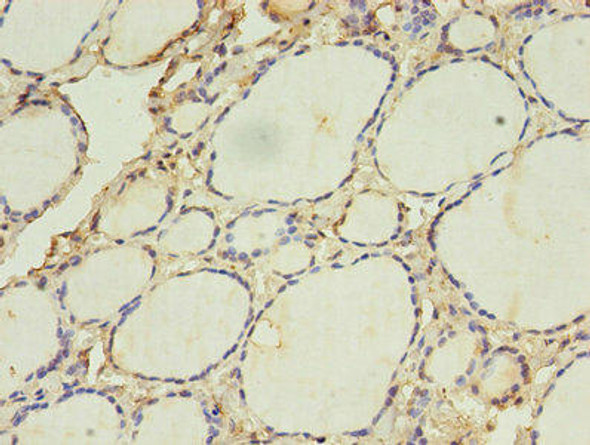
| Background: | Probable core component of the endosomal sorting required for transport complex III (ESCRT-III) which is involved in multivesicular bodies (MVBs) formation and sorting of endosomal cargo proteins into MVBs. MVBs contain intraluminal vesicles (ILVs) that are generated by invagination and scission from the limiting membrane of the endosome and mostly are delivered to lysosomes enabling degradation of membrane proteins, such as stimulated growth factor receptors, lysosomal enzymes and lipids. The MVB pathway appears to require the sequential function of ESCRT-O, -I,-II and -III complexes. ESCRT-III proteins mostly dissociate from the invaginating membrane before the ILV is released. The ESCRT machinery also functions in topologically equivalent membrane fission events, such as the terminal stages of cytokinesis. Together with SPAST, the ESCRT-III complex promotes nuclear envelope sealing and mitotic spindle disassembly during late anaphase. Plays a role in the endosomal sorting pathway. ESCRT-III proteins are believed to mediate the necessary vesicle extrusion and/or membrane fission activities, possibly in conjunction with the AAA ATPase VPS4. When overexpressed, membrane-assembled circular arrays of CHMP4B filaments can promote or stabilize negative curvature and outward budding. CHMP4A/B/C are required for the exosomal release of SDCBP, CD63 and syndecan. |
| Synonyms: | Charged multivesicular body protein 4b (Chromatin-modifying protein 4b) (CHMP4b) (SNF7 homolog associated with Alix 1) (SNF7-2) (hSnf7-2) (Vacuolar protein sorting-associated protein 32-2) (Vps32-2) (hVps32-2), CHMP4B, C20orf178 SHAX1 |
| UniProt Protein Function: | CHMP4B: Probable core component of the endosomal sorting required for transport complex III (ESCRT-III) which is involved in multivesicular bodies (MVBs) formation and sorting of endosomal cargo proteins into MVBs. MVBs contain intraluminal vesicles (ILVs) that are generated by invagination and scission from the limiting membrane of the endosome and mostly are delivered to lysosomes enabling degradation of membrane proteins, such as stimulated growth factor receptors, lysosomal enzymes and lipids. The MVB pathway appears to require the sequential function of ESCRT-O, -I,-II and -III complexes. ESCRT-III proteins mostly dissociate from the invaginating membrane before the ILV is released. The ESCRT machinery also functions in topologically equivalent membrane fission events, such as the terminal stages of cytokinesis and the budding of enveloped viruses (HIV-1 and other lentiviruses). ESCRT-III proteins are believed to mediate the necessary vesicle extrusion and/or membrane fission activities, possibly in conjunction with the AAA ATPase VPS4. When overexpressed, membrane-assembled circular arrays of CHMP4B filaments can promote or stabilize negative curvature and outward budding. Via its interaction with PDCD6IP involved in HIV-1 p6- and p9-dependent virus release. Defects in CHMP4B are the cause of cataract posterior polar type 3 (CTPP3). A subcapsular opacity, usually disk-shaped, located at the back of the lens. It can have a marked effect on visual acuity. Belongs to the SNF7 family. |
| UniProt Protein Details: | Protein type:Membrane protein, peripheral Chromosomal Location of Human Ortholog: 20q11.22 Cellular Component: internal side of plasma membrane; membrane coat; late endosome membrane; cytoplasm; midbody; nucleus; cytosol; endosome Molecular Function:identical protein binding; protein binding; protein homodimerization activity Biological Process: cell separation during cytokinesis; viral reproduction; vacuolar transport; non-lytic virus budding; regulation of viral reproduction; viral infectious cycle; posttranslational protein targeting to membrane; endosome transport; mitotic metaphase plate congression; nuclear organization and biogenesis; protein homooligomerization Disease: Cataract 31, Multiple Types |
| NCBI Summary: | This gene encodes a member of the chromatin-modifying protein/charged multivesicular body protein (CHMP) protein family. The protein is part of the endosomal sorting complex required for transport (ESCRT) complex III (ESCRT-III), which functions in the sorting of endocytosed cell-surface receptors into multivesicular endosomes. The ESCRT machinery also functions in the final abscisson stage of cytokinesis and in the budding of enveloped viruses such as HIV-1. The three proteins of the CHMP4 subfamily interact with programmed cell death 6 interacting protein (PDCD6IP, also known as ALIX), which also functions in the ESCRT pathway. The CHMP4 proteins assemble into membrane-attached 5-nm filaments that form circular scaffolds and promote or stabilize outward budding. These polymers are proposed to help generate the luminal vesicles of multivesicular bodies. Mutations in this gene result in autosomal dominant posterior polar cataracts.[provided by RefSeq, Oct 2009] |
| UniProt Code: | Q9H444 |
| NCBI GenInfo Identifier: | 24636296 |
| NCBI Gene ID: | 128866 |
| NCBI Accession: | Q9H444.1 |
| UniProt Secondary Accession: | Q9H444,Q53ZD6, E1P5N4, |
| UniProt Related Accession: | Q9H444 |
| Molecular Weight: | 224 |
| NCBI Full Name: | Charged multivesicular body protein 4b |
| NCBI Synonym Full Names: | charged multivesicular body protein 4B |
| NCBI Official Symbol: | CHMP4B |
| NCBI Official Synonym Symbols: | SNF7; CTPP3; Shax1; CHMP4A; SNF7-2; VPS32B; CTRCT31; Vps32-2; C20orf178; dJ553F4.4 |
| NCBI Protein Information: | charged multivesicular body protein 4b; hSnf7-2; hVps32-2; chromatin modifying protein 4B; chromatin-modifying protein 4b; SNF7 homolog associated with Alix 1; Snf7 homologue associated with Alix 1; vacuolar protein sorting-associated protein 32-2; vacuolar protein-sorting-associated protein 32-2 |
| UniProt Protein Name: | Charged multivesicular body protein 4b |
| UniProt Synonym Protein Names: | Chromatin-modifying protein 4b; CHMP4b; SNF7 homolog associated with Alix 1; SNF7-2; hSnf7-2; Vacuolar protein sorting-associated protein 32-2; Vps32-2; hVps32-2 |
| Protein Family: | Charged multivesicular body protein |
| UniProt Gene Name: | CHMP4B |
| UniProt Entry Name: | CHM4B_HUMAN |